Application release V2.1 launch date is here
After listening to users and accreditation bodies and learning from our own experiences interacting with the application, we have been hard at work on the latest upgrade to R18LabQMS – Version 2.1.
Our focus on this upgrade was to implement a more intuitive design in areas that were the most challenging, which will allow the user to more easily navigate those areas.
The soft launch date for V2.1 is now set for Wednesday, April 8th at 2 p.m. EDT. Please see the details under “Application News” on the login page for all news and updates.
Let’s take a look at V2.1 upgrades
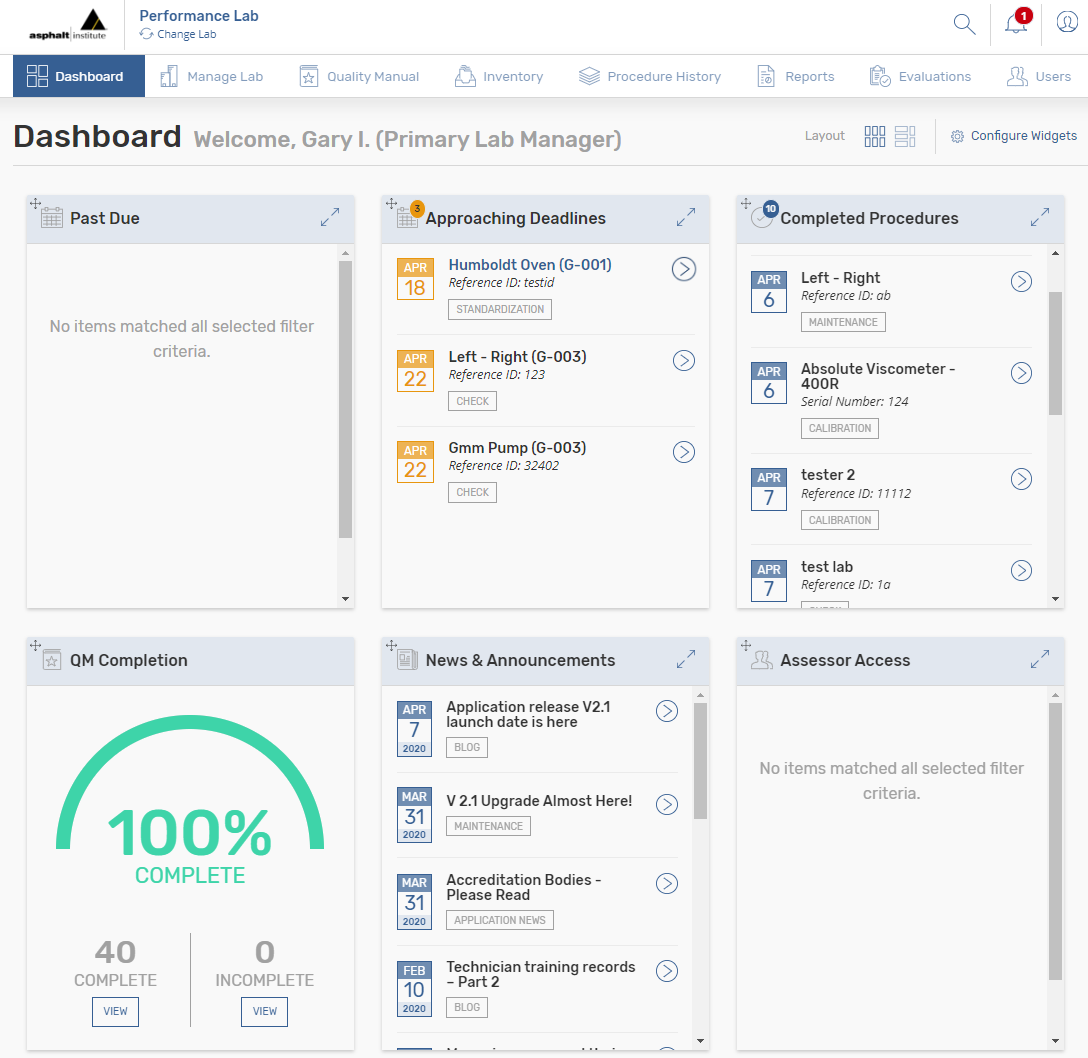
1) Alert bell
Notifications are located at the top right corner of the screen where you will find the alert bell icon. The number of notifications at any given time will appear within a red circle on the bell, while all notifications including “Equipment Procedures” are listed under the bell. This list can become quite lengthy and the only interaction is to either click “Refresh List” or “Mark All As Read” – which changes the color of the notification from red to black. This action is tedious and has been addressed in V2.1.
>> V2.1 Upgrade for the alert bell
We have eliminated all equipment related procedures on the alert bell and only include more relevant items that are not otherwise addressed within the application, i.e., technician evaluations and certifications, QM completion, etc. Once the user clicks the alert bell, there will be a list of items that can be individually addressed by clicking view. The other option is to dismiss individual items as you review the list. The last option is at the top of the list, clear all notifications. When you do, there will be a warning message that reads, “This action will remove all current notifications from your account forever. Are you sure you want to do that?”
2) Search
The search mechanism is located in the top right-hand corner of the screen where you will find a magnifying glass icon. Currently, when performing a search within the system a white space appears with the results not being specific and a list that some find long and confusing.
>> V2.1 Upgrade for search
With the upgrade, you will be able to perform a search function and get more specific results. For example: if you put in a specific equipment serial number, one result would come up with that specific piece of equipment.
3) Add Links to AASHTO, ASTM
Within the application, there’s a test methods widget within the “Manage Lab” tab that lists most, if not all, of the construction materials tests for ASTM and AASHTO Standards (state and international standards can be added). This information is used throughout the application, namely when creating technician training records. One of the most frequently asked questions is, “Can we click on the standard designation?” The answer is always, “no”, because of copyrights it’s only available via subscription or purchasing a hard copy.
>> V2.1 Upgrade for add Links to AASHTO, ASTM
In V2.1 we created a link to both ASTM and AASHTO at the top of each column of standard designations that will take you to their respective log-in pages for users that subscribe to their services for online standards.
4) Last technician evaluation, recall, edit, save
Creating a new technician evaluation is one of the most important parts of maintaining consistent quality and accreditation. This requires good record-keeping and is done within the application by going to the “Evaluations” tab and following the prompts at the top. Once an evaluation record has been completed the process is repeated on a periodic basis, typically 12 months.
>> V2.1 Upgrade for last technician evaluation, recall, edit, save
The procedure of creating new technician evaluations can be a long process, especially if there are multiple training records with several technicians. With the new upgrade, you will be able to recall the last evaluation record for each technician, indicate the evaluation date for each record and add or delete a training record. This process will be more efficient and save an enormous amount of time.
5) QMS training records accordion
All training records are found in the Quality Manual within section 5.5.4. Currently, when records are completed, they are listed individually. With a single technician having several records and multiplying this by the number of technicians in the lab, this list can get quite lengthy over several years.
>> V2.1 Upgrade for QMS Training records accordion
In the upgrade, each user (technician) that has a completed training record will automatically have their name listed in section 5.5.4. The number indicated on the far right will be the number of completed technician evaluations. Click on a name and the evaluations for that technician will expand down with a complete listing – like an accordion.
6) Signature line on procedures
Currently, when completing an equipment procedure, one of the required fields is signature. However, the person completing the procedure only has the option to type in their name. This has occasionally been brought up about an actual (digital) signature. While this meets the criteria of the accreditation body some companies require an actual signature.
>> V2.1 Upgrade for signature line on procedures
We have addressed this on V2.1 with the ability to provide an actual signature on all equipment procedure forms. This is done by the user providing a signature on a document and uploading it to their specific profile. Find the profile link on the upper right-hand portion, click and “Add a Signature.”
Going forward: When completing procedures you will have the option to just type in your name as it is now or to click the box ‘Sign Using Signature Image’ to use your actual signature. A printed version of the user’s name and title will automatically accompany the signature image directly below the signature box.
7) Equipment table – procedures
The most detailed part of the application is creating and maintaining equipment procedures. Currently, the only way to see if there are procedures associated with equipment is to individually click “view” and then you need to know where to look. It’s easy to miss and verify that all equipment procedures are in the system.
>> V2.1 Upgrade for equipment table – procedures
The solution: When going to the “Inventory” tab, there are now 2 sub-tabs: “Lab Equipment” and “Equipment Procedures.” The “Equipment Procedures” tab has not changed as it currently is, however when clicking the “Equipment Procedures” tab, you are now able to view all procedures associated with your equipment inventory. Additionally, you will be able to edit, see details, or perform procedures from this page view. This will be a game-changer for the users to see the big picture with the procedures they have in place.
User questions
Q: The users that we have added to R18LabQMS didn’t receive the email to create their password, what do we do now?
A: Great question! First, I’ll walk through the process of adding a user, their user ID and password. When adding a user they will immediately receive an email from service@r18labqms.com. It’s important for that user to check their email as soon as they have been added to their account. If they don’t find it, check their clutter or deleted folders, if that fails it’s possible that the IT Department is blocking certain domains. Once the new user receives the email there will be a link that will take him/her to a page to create a password and once the password is created they will be able to login into the application.
The Primary Lab Manager can resend the email to the new user if needed by first going to the “Users” tab, click “View” on the right-hand column, and then click “Resend Email Confirmation.” Please note that if a user has already created a password, the “Resend Email Confirmation” will not appear.
Resources and training opportunities
Once you work through the setup process, the following links can be found in the lower right-hand corner of the application:
• Support
• User Guide
• R18 Blog
• WebEx – Additional training, please let us know
• Onsite Training – Fee-based, please inquire
Upcoming Meetings
AAPT (Association of Asphalt Paving Technologist)
• Sponsor (on-site)
• Tentative dates are now September 13-16, 2020, San Diego, CA
SEAUPG 2020 Annual Meeting & Exhibits
• Sponsor (on-site)
• November 2020
For all questions regarding your quality management system, whether your lab is accredited or not, please contact us and learn how R18LabQMS can add value to your company.
Gary Irvine
R18LabQMS Program Manager